Plastic photodegradation and chemical breakdown
In this newsletter we briefly and shortly discuss important subtopics like;
- Premium regrinds, virgin resins and nature conditions
- Direct sunlight and greenhouse prevention benefits
- Capturing fugitive gases and End-of-Waste benefits
- Fume hazards due to off-gassing
- The quality of fibre components in regeneration
- Photodegradation effects on polymers
- The fibre base in polymers
- Photodegradation and monomerization of polymers
LISTEN TO THIS NEWSLETTER WITH AN AUGMENTED CONVERSATION. PLAY THE AUDIOCAST LINK Plastic photodegradation and chemical breakdown

This explores the repurposing potential of premium regrinds for high-grade applications and the financial rewards of lifecycle maximization through proper storage, while detailing further economic gains from achieving zero-waste status by capturing gaseous vapours as a resource. We also examine the off-gassing, auto-oxidation, and aromatic properties of polymer and semi-polymer materials, alongside the dual impacts of photodegradation on resource weight loss and carbon fibre chain scission. Finally, the edition compares the effects of photocatalysis across different material types, evaluates the resilience and byproducts of their fibre bases, and analyses the role of high-energy solar radiation in reducing the size of persistent plastic waste.
BASED ON CINTRA 2021 ARTICLE Photocatalytic Degradation of Plastic Waste: A Mini Review
PREMIUM REGRINDS, VIRGIN RESINS AND NATURE CONDITIONS

Premium regrind made with the best quality waste material and most advanced environmental processes is less durable than virgin resin
This is because its “molecular clock” has already started. No matter how clean the material is, the heat and stress of its first life have permanently altered its chemistry.
Here is the “why” in four points:
Weakened Structure: The polymer chains are physically shorter (chain scission), meaning they tangle less and crack more easily under pressure.
Depleted Defences: The original protective additives (antioxidants and UV inhibitors) were “used up” during the first round of processing and use.
UV Sensitivity: Previous exposure creates “UV-hungry” chemical sites (chromophores) that cause the plastic to yellow and become brittle much faster than new material.
Micro-Flaws: Minor impurities and batch variations create “stress points” that lead to premature failure when the plastic is exposed to the elements.
The Bottom Line:
To make regrind behave like virgin resin, you have to “upcycle” it by chemically replenishing the stabilizers and modifiers it lost.
There are wide economic and structural benefits of protecting polymers like PP, PS, and PVC from UV light and heat. By shielding these materials, you effectively “stop the clock” on their degradation
1. Structural Preservation
- Chain Integrity: Avoiding UV and heat prevents “chain scission,” keeping polymer chains long and strong. This maintains the material’s original tensile strength and flexibility.
- Preventing “Chalking”: Proper storage stops fibres from becoming brittle or turning into low-value dust, keeping the material viable for high-grade reuse.
2. Economic Value & Marketability
- Colour Versatility: Preventing yellowing (chromophore formation) ensures the recycled material can be dyed into a wider range of colours, increasing its resale options.
- Premium Pricing: “Clean” waste with minimal thermal history commands a higher market price because it is easier to process and closer to virgin resin quality.
3. Mitigating “Auto-Acceleration”
- Heat Loops: Trapped heat (the greenhouse effect) creates a feedback loop that accelerates chemical breakdown.
- Mass Retention: Cool, dark storage prevents the loss of material weight through off gassing (VOCs), ensuring you have more physical product to sell or reuse.
4. Financial & Environmental ROI
- ESG Benefits: Longer material lifecycles reduce carbon footprints, potentially qualifying companies for tax incentives or carbon credits.
- Lower Costs: Processors save money because non-degraded material requires less energy and fewer expensive “restorative” additives to upcycle.
DIRECT SUNLIGHT AND GREENHOUSE PREVENTION BENEFITS

CAPTURING FUGITIVE GASES AND END-OF-WASTE BENEFITS

Capturing and treating plastic off gases flips the script from an environmental liability to a massive economic win
Turning Waste into Revenue
- Chemical Recovery: Scrubbing systems can capture byproduct gases and convert them into sellable industrial goods, such as Hydrochloric Acid (HCl) from PVC or high-purity styrene monomers from Polystyrene.
- Energy Generation: Once cleaned of impurities, captured gases (Syngas) can be burned as a low-carbon fuel to power the facility, slashing utility bills.
Regulatory & Financial Shielding
- Tax Incentives: Facilities that capture toxic VOCs can often earn carbon credits or benefit from reduced pollution taxes, directly padding the bottom line.
- Fine Avoidance: Effective treatment ensures compliance with strict emission laws (like the Industrial Emissions Directive), preventing heavy fines and legal fees.
Asset & Catalyst Protection
- Corrosion Control: Acidic gases like HCl eat through metal piping and machinery. Scrubbing these gases extends the lifespan of expensive infrastructure, deferring major capital expenditures (CAPEX).
- System Longevity: Removing contaminants prevents the “poisoning” of specialized catalysts like TiO2, keeping the chemical processes efficient for longer periods.
Premium Market Positioning
- ESG Value: A “zero-emission” closed-loop process attracts ESG-conscious investors and secures high-value corporate contracts.
- High-Quality Recyclate: Decontaminated, odor-free plastics reach “End-of-Waste” status, allowing them to be sold at a premium for sensitive uses like food packaging or medical devices.
Heat and photocatalysts trigger the chemical breakdown of common plastics, leading to the release of specific, often hazardous, volatile organic compounds (VOCs). The type of “fume” produced is a direct map of the plastic’s molecular structure
1. Polyvinyl Chloride (PVC): Acidic & Corrosive
- Main Byproduct: Hydrogen Chloride (HCl) gas, which turns into hydrochloric acid upon contact with moisture (eyes/lungs).
- Toxic Risks: Intense heat can produce Dioxins and Furans, which are highly toxic even in trace amounts.
- Aromatics: As the chain “unzips,” it can form Benzene and Toluene.
PVC is the most chemically aggressive when heated due to its chlorine content.
2. Polystyrene (PS): Aromatic Irritants
- Main Byproduct: Styrene Monomer gas, a respiratory irritant and potential carcinogen.
- Odor: Auto-oxidation creates Benzaldehyde and Acetophenone, giving degrading foam a sickly-sweet smell.
- Pollutants: High temperatures can fuse these rings into Polycyclic Aromatic Hydrocarbons (PAHs).
Because PS contains a benzene ring on every other carbon, its off gassing is dominated by “styrenics.”
3. Polypropylene (PP): Pungent Aliphatics
- Main Byproducts: Formaldehyde and Acetaldehyde, which are pungent, irritating aldehydes.
- Acrid Fumes: Produces Acrolein, a notoriously sharp-smelling irritant.
- Random Scission: Releases a mixture of acetone, acetic acid, and volatile alkanes (like ethane).
Lacking aromatic rings, PP breaks into straight or branched chain fragments.
4. The Catalyst Effect
- Higher Oxygenation: You get more carbon monoxide, alcohols, and carboxylic acids rather than simple hydrocarbons.
- The “Middle” Stage: While the goal is to break everything down into harmless CO2 and water, the material often “off-gasses” volatile intermediates (like aldehydes) before they can be fully neutralized.
Adding a photocatalyst like Titanium Dioxide lowers the “energy bar” for these reactions to happen.
FUME HAZARDS DUE TO OFF GASSING

THE QUALITY OF FIBRE COMPONENTS IN REGENRATION

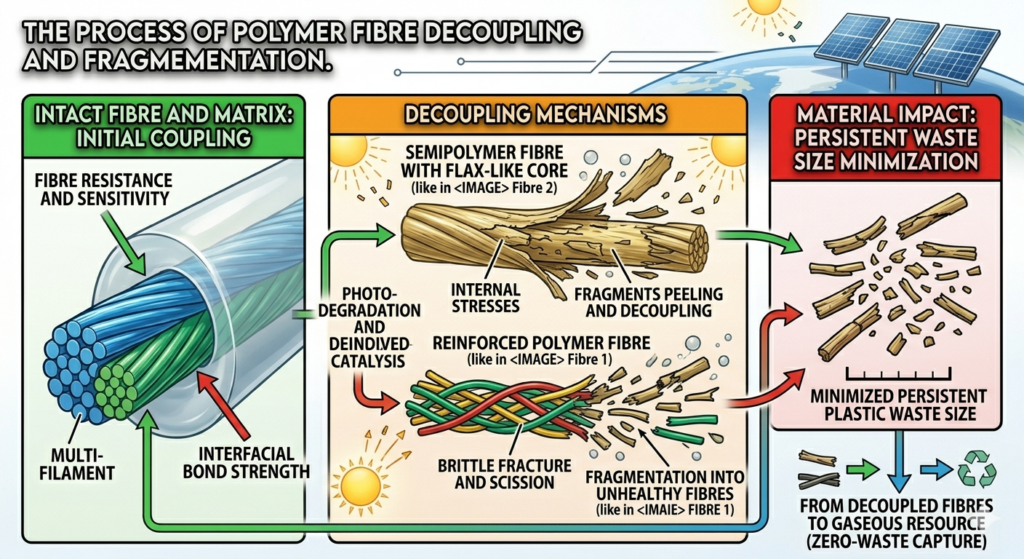
Photodegradation acts as a “double-edged sword” for plastic recycling—it can either ruin the material’s physical strength or provide a clever chemical shortcut to high-quality reuse
Mechanical Decline: The “Short Chain” Problem
- Molecular Loss: UV light breaks long polymer chains into smaller fragments (chain scission). Since chain length equals strength, this makes the material brittle.
- The “Virgin” Gap: To reuse this degraded plastic in new products, manufacturers usually have to mix in expensive “virgin” resin to compensate for the lost durability.
Physical Damage: Surface Micro-Cracking
- Stress Points: Photodegradation starts at the surface, creating microscopic pits and cracks.
- Failure Risks: In fibres like Polypropylene (PP), these tiny cracks act as “snap points.” Once a fibre is pitted, it can no longer be used for high-end textiles because it will break under even minor tension.
Chemical “Pollution”: Oxygenated Groups
- Hidden Defects: The process introduces new chemical groups (carbonyls and hydroperoxides) into the plastic’s backbone.
- Processing Hurdles: These groups act as impurities that cause yellowing and bad odors (off-gassing) when the plastic is melted down for repurposing.
The Strategic Flip: Chemical “Unzipping”
- Monomer Recovery: By using catalysts to intentionally target specific bonds, you can “zip down” the plastic into its original building blocks (monomers).
- High-Quality Output: This allows for the creation of “like-new” plastic, completely bypassing the weakness and yellowing issues of standard recycled flakes.
Polyesters (like PET) and carbon-chain polymers (like PP, PS, and PVC) react so differently to sunlight. The “weak point” of the plastic determines how fast it fails and how easily it can be recycled
1. The “Snap” vs. the “Grind”: Bond Strength
- Carbon-Chains (PP, PS, PVC): These have a backbone of Carbon-Carbon bonds. Breaking them is a slow, messy process called auto-oxidation. It requires a “free radical” to attack the chain repeatedly to cause damage.
- Polyesters (PET): These have ester bonds built right into the spine. UV light can trigger a Norrish cleavage, which acts like a pair of chemical scissors, snapping the backbone much more directly than the radical process used on carbon chains
2. Resilience and Structural Defense
- The “Weakest” (PP): Polypropylene is notoriously UV-sensitive. Its “tertiary carbons” are easy targets for radicals, causing it to turn into white powder (chalking) very quickly in the sun.
- The “Shielded” (PET): PET contains aromatic rings that can absorb and “spread out” UV energy, giving it better natural resistance than PP. However, once it hits its limit, it undergoes “phototendering,” where it yellows and loses its pull-strength (tensile strength).
3. Upcycling vs. Destruction
- Polyolefins (Messy Breakdown): When PP or PS break down, they create a random mixture of hydrocarbons. This makes it hard to turn them back into “new” plastic; the goal is usually to just break them down into CO2 and water.
- Polyesters (Clean Breakdown): Because UV specifically targets the ester bonds, PET is a “gold mine” for chemical recycling. Instead of just burning it or burying it, scientists can use light to “snip” the chains back into high-value building blocks (like carboxylic acids) to create brand-new, virgin-quality plastic.
PHOTODEGRADATION EFFECTS ON POLYMERS

THE FIBRE BASE IN POLYMERS
PP, PVC, and PS are all common plastics, Polypropylene (PP) is the only one that functions effectively as a structural fibre.
Polypropylene (PP): The Fiber Specialist
PP is the “steel of plastics” in the fibre world because of its high tensile strength and durability.
- The Process: It is created via melt spinning, where melted resin is forced through tiny holes to create long filaments.
- Unique Property: It is hydrophobic (repels water), making it ideal for sportswear and thermal clothing.
- Use Cases: Beyond textiles, it is used in concrete reinforcement to prevent cracking, and in industrial ropes and medical masks.
Polyvinyl Chloride (PVC): The Coating Material
PVC isn’t typically spun into a stand-alone fibre; instead, it acts as a “shield” for other fabrics.
- The Hybrid Approach: PVC is usually coated over a base of polyester or nylon.
- Use Cases: This creates a heavy-duty, waterproof layer used for industrial tarpaulins, rainwear, and upholstery.
Polystyrene (PS): The Rigid Outlier
PS is unsuitable for fibres because its molecular structure is too brittle; a PS fibre would snap rather than bend.
- Forms: It is strictly used in rigid shapes (like petri dishes) or expanded foam (like shipping coolers).
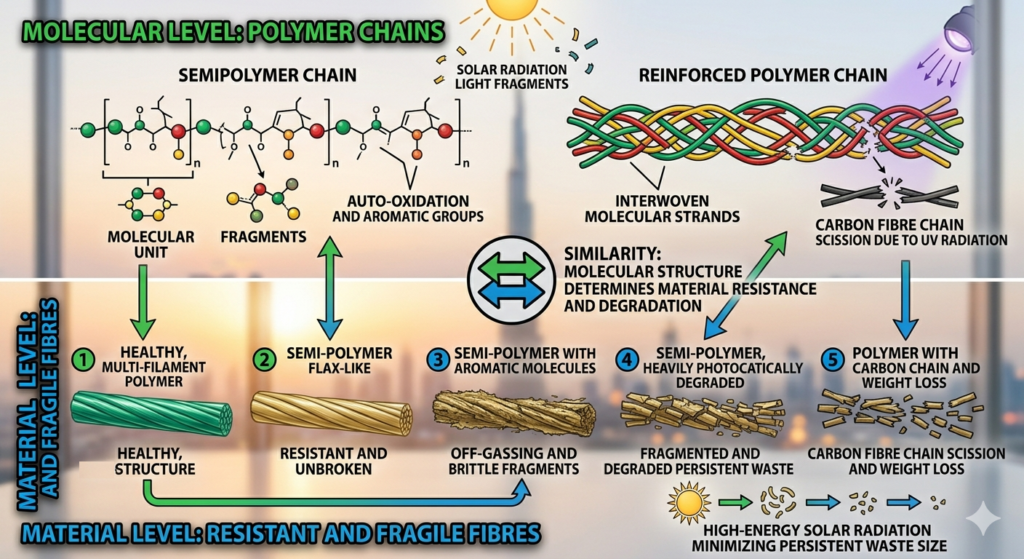
Photodegradation acts as a high-speed “deconstruction crew” for plastics, using light and chemistry to turn long-lasting waste into simpler, smaller components.
1. Breaking the Backbone (Chain Scission)
The core of the process is chain scission, which physically cuts the long, sturdy polymer chains into tiny fragments. This drastically reduces the material’s molecular weight, turning a strong plastic into a weak, degraded substance that is much easier to break down further.
2. Using Catalysts as Accelerators
Left alone, most plastics could take centuries to decompose. By adding semiconductors like Titanium Dioxide, solar energy is harnessed to “fast-forward” this timeline. These catalysts lower the energy needed to start the breakdown process, making decomposition happen in a fraction of the time.
3. The Free Radical “Chain Reaction”
High-energy UV light strikes the plastic to break C-C and C-H bonds, creating highly reactive “free radicals.” This kicks off an auto-oxidation loop:
- Propagation: These radicals react with oxygen and neighboring polymer chains.
- Self-Destruction: The reaction spreads through the material like a wildfire, causing continuous breaking of the molecular structure.
4. Chemical Mineralization (ROS)
Photocatalysts create Reactive Oxygen Species (ROS), such as hydroxyl radicals. These are chemically aggressive enough to “eat” the organic fragments, eventually turning them into harmless (a process called mineralization).
5. Enhanced Efficiency
In specific materials like Polystyrene (PS), adding creates a composite that loses weight and molecular integrity significantly faster than pure plastic. This proves that intentional photodegradation is a highly effective tool for tackling persistent plastic waste.
PHOTODEGRADATION AND MONOMERIZATION OF POLYMERS